Acids Bases Ph Worksheet Answers: A Comprehensive Guide
Understanding Acids and Bases
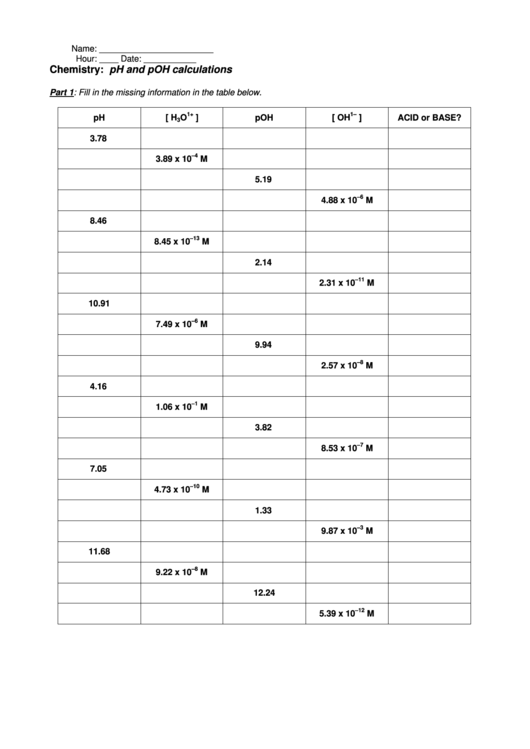
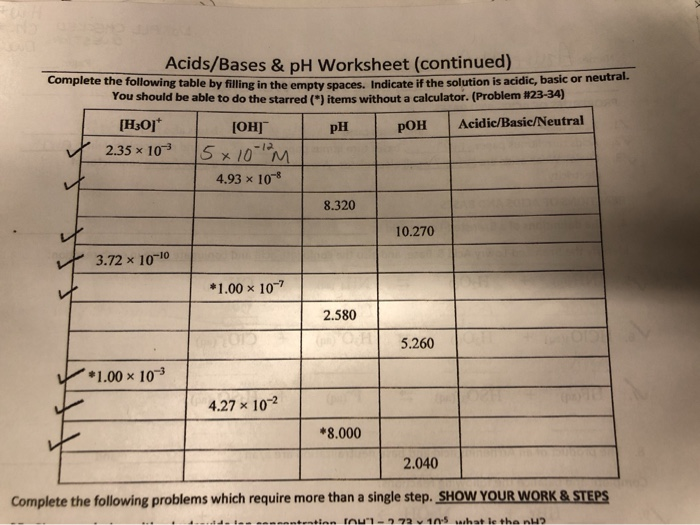
Acids and bases are fundamental concepts in chemistry, and understanding their properties and behaviors is crucial for students and professionals alike. When it comes to acids and bases, one of the most important concepts to grasp is pH levels. pH is a measure of the concentration of hydrogen ions in a solution, and it can range from 0 to 14. A pH of 7 is considered neutral, while a pH below 7 is acidic and above 7 is basic.
For students working on an acids bases ph worksheet, it's essential to have a solid understanding of the basics. This includes knowing the definitions of acids and bases, understanding the pH scale, and being able to calculate pH levels. With this knowledge, students can tackle more complex problems and exercises, such as identifying the pH of a solution, determining the strength of an acid or base, and solving problems involving acid-base reactions.
Calculating pH Levels
To solve acids bases ph worksheet problems, students need to understand the different types of acids and bases, including strong and weak acids and bases. Strong acids and bases completely dissociate in water, while weak acids and bases only partially dissociate. This understanding is critical for calculating pH levels and solving problems involving acid-base reactions. Additionally, students should be familiar with the different methods for calculating pH, including using pH formulas and tables.
With a solid understanding of acids and bases, students can move on to more advanced topics, such as calculating pH levels. This involves using formulas and tables to determine the pH of a solution. By practicing with acids bases ph worksheet answers, students can develop their problem-solving skills and become more confident in their ability to calculate pH levels. Whether you're a student looking for help with your homework or a professional seeking to review the basics, our comprehensive guide to acids bases ph worksheet answers has got you covered.