Printable Periodic Table of Electronegativity: A Comprehensive Guide
Understanding Electronegativity
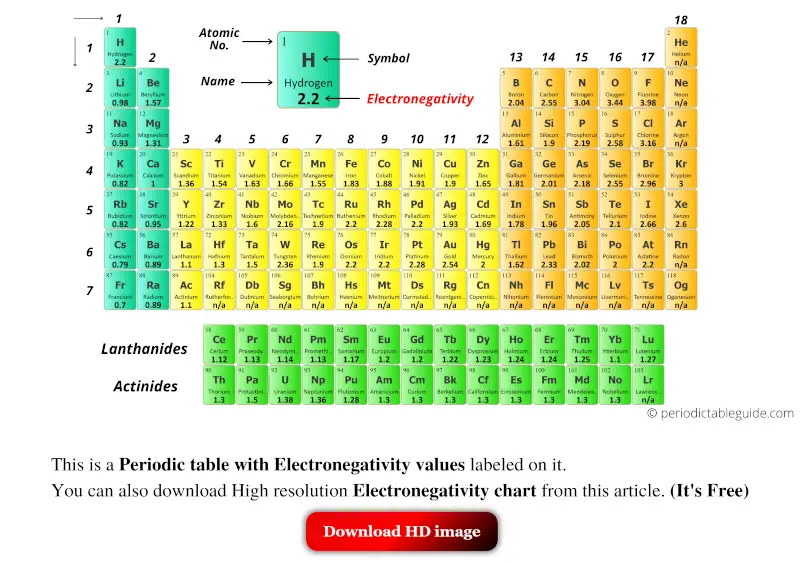
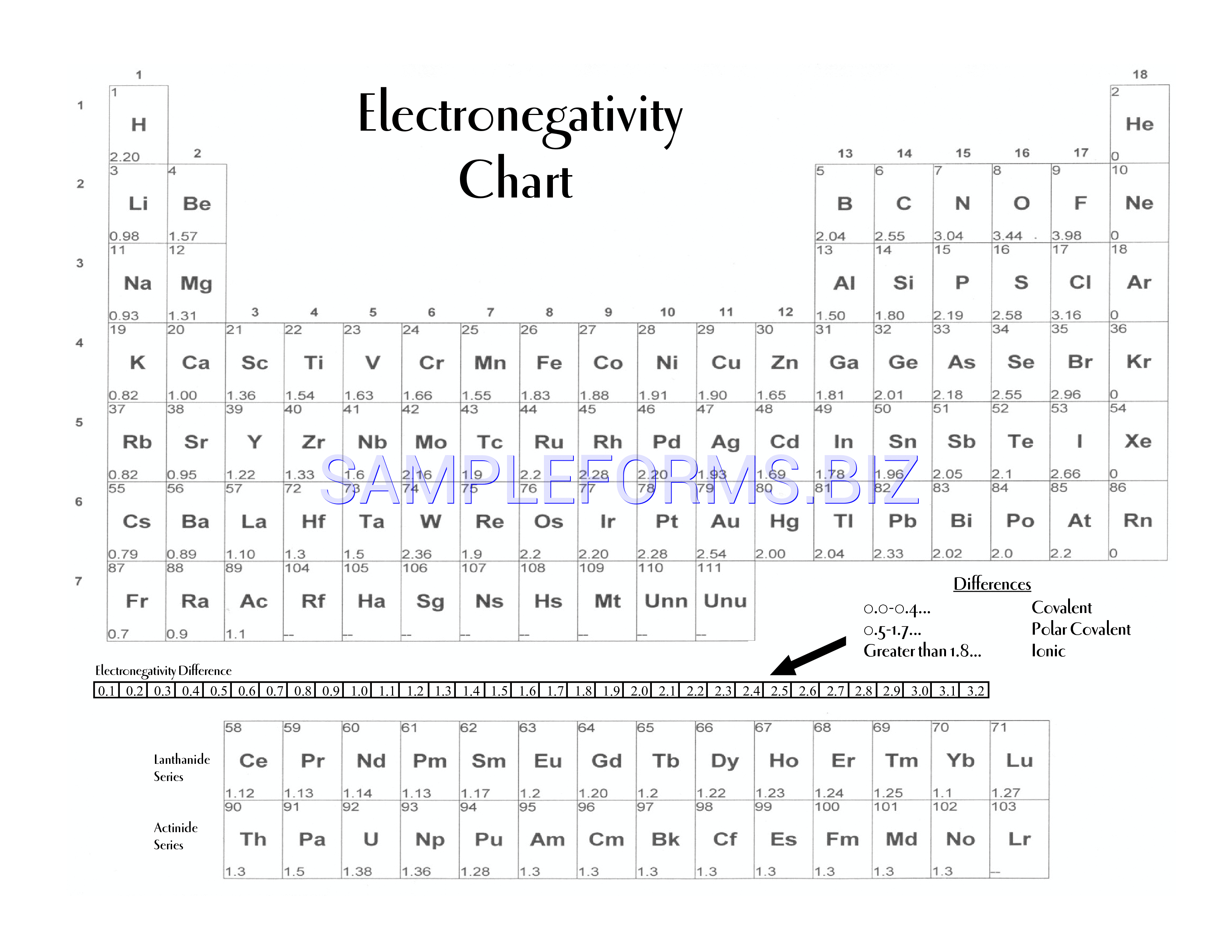
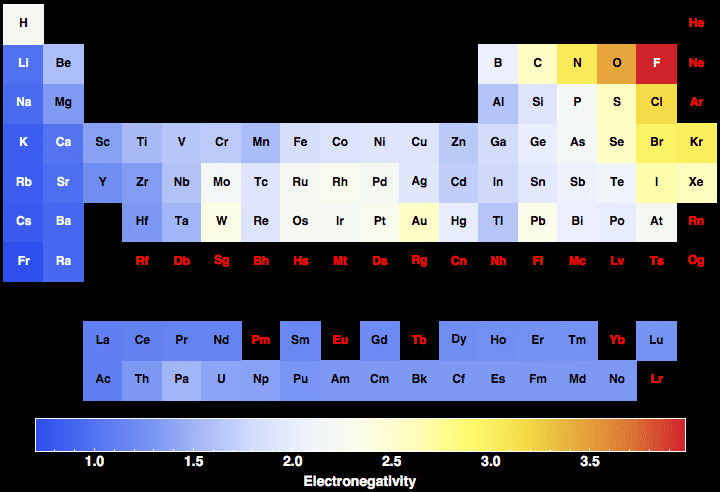
The periodic table is a fundamental tool in chemistry, and understanding the concept of electronegativity is crucial for students and professionals alike. Electronegativity refers to the ability of an atom to attract electrons in a covalent bond. The printable periodic table of electronegativity provides a visual representation of the elements and their corresponding electronegativity values, making it easier to compare and analyze the properties of different elements.
The electronegativity of an element is measured on the Pauling scale, which ranges from 0 to 4.0. Fluorine has the highest electronegativity value of 4.0, while cesium has the lowest value of 0.79. The printable periodic table of electronegativity allows users to quickly identify the electronegativity values of different elements and understand how they interact with each other.
Benefits of a Printable Periodic Table
Electronegativity plays a critical role in determining the properties of molecules and compounds. By understanding the electronegativity values of different elements, chemists can predict the polarity of bonds, the reactivity of elements, and the stability of compounds. The printable periodic table of electronegativity is an essential resource for anyone studying chemistry, from high school students to professional researchers.
A printable periodic table of electronegativity offers several benefits, including easy access to information, improved retention, and enhanced visualization. By having a physical copy of the periodic table, users can quickly reference the electronegativity values of different elements and make notes or annotations as needed. Additionally, the visual representation of the elements and their electronegativity values can help users to better understand the relationships between different elements and their properties.